LEADERSHIP
MESSAGE

Mark Kessel,
CHAIRMAN OF THE BOARD OF DIRECTORS

Bill Rodriguez,
CHIEF EXECUTIVE OFFICER
While COVID-19 continued to wreak havoc on people and communities throughout 2021, the year was in many ways a year of acceptance. Acceptance that COVID-19 has changed the world forever—from work practices to travel, from schooling to sport, from providing care to receiving care. Acceptance that health is not something that happens outside of the economy, but that health and economic activity are inextricably intertwined, and not just for pandemic viruses. Acceptance, ultimately, that spending on healthcare is truly an investment, not just an outlay.
“Build back better” was the catchphrase of the year, and certainly in diagnostics a lot of progress has been made in pursuit of better, more sustainable, more resilient health systems the world over. From building new manufacturing facilities in Africa, India and Latin America; to accelerating the development of a new generation of point-of-care tests for primary healthcare; to making available inexpensive self-tests that can help people protect themselves and their families; to powering up new genome sequencing machines that detect new variants as they emerge—investments made in diagnostics for the pandemic are indeed building a legacy that will have far-reaching impact beyond COVID-19.
Activities in relations to other diseases continued to experience disruption in 2021. The indefatigable commitment of our colleagues and partners meant we were able to achieve some important milestones, including the generation of data that were urgently needed to inform WHO guidelines on hepatitis C testing, and major progress in tuberculosis diagnostic technologies, including CAD-based imaging and next-generation LAM testing. New digital tools are in development with potential applicability to many diseases. We were also able to make strides in our fledgling programme in non-communicable diseases, notably by achieving significant price reductions for diabetes test strips and launching a new initiative to improve cervical cancer testing and screening using innovative technologies to help meet new WHO targets.
FIND’s current strategy, published in 2021, focuses on diagnostic testing as an enabler of both universal health coverage and global
health security and calls on our partners and stakeholders to support a global alliance that can accelerate diagnostic progress, from innovation to access. This call was amplified by The Lancet Commission on Diagnostics, whose report we helped launch in October 2021.
However, for all the things that have been achieved, one thing that is not changing fast enough is equitable access to healthcare. Delays in the availability and uptake of COVID-19 tests and vaccines, most pronounced in low- and middle-income countries, have directly contributed to the emergence and global spread of the Delta and Omicron variants, extending the pandemic into its third year. Even as many high-income countries are accepting that COVID-19 is now a fact of life and are using the tools and resources available to them to promote full economic activity, with our Access to COVID-19 Tools (ACT) Accelerator partners we are not willing to accept that COVID-19 should continue to threaten the health, education and welfare of the poor and vulnerable.
With this 2021 report, we celebrate accomplishments in 2021, and we acknowledge how much work remains to be done—not only to bring the pandemic to an end, but to defeat so many other diseases that continue to claim countless lives and jeopardize livelihoods every single day. We offer heartfelt thanks to the FIND team, and to all our friends, colleagues, partners, and stakeholders across the world for your ongoing support, as we continue to work together in our mission to ensure equitable access to reliable diagnosis around the world.

PRIORITIZING COLLABORATION AND EQUITY IN ADDRESSING GLOBAL HEALTH THREATS
Diagnostics are fundamental to sustainable, resilient and equitable health systems capable of navigating global health emergencies. From helping stop the spread of COVID-19 and informing public health decisions, to measuring the effectiveness of vaccines and treatments and detecting new variants—global collaboration and equity are key to ensure all countries can rapidly detect and contain any new health threat.

SEE HOW WE PARTNERED WITH THSTI TO EXPAND COVID-19 TESTING CAPACITY DURING INDIA'S SECOND WAVE
"Rapid scale-up of testing capacity for COVID-19 relied on global collaboration and high-speed innovation, from R&D to implementation. By working with partners and local communities across the full value chain, we have been able to address key barriers to testing, such as quality issues, cost and geographic distance. Our work as part of the ACT-Accelerator continues to support the deployment of tailored testing strategies and decentralized testing models that are playing a critical role in getting the pandemic under control."
EMMA HANNAY, CHIEF ACCESS OFFICER
INTRODUCING AND SCALING CRITICAL NEW COVID-19 DIAGNOSTIC TESTS FOR USE IN LOW- AND MIDDLE-INCOME COUNTRIES (LMICs)


The global community continued to rally around the Access to COVID-19 Tools (ACT) Accelerator, which created unprecedented impact in 2021—expanding its operational reach into over 50 countries on six continents. As ACT-Accelerator Diagnostics Pillar co-convener, FIND worked at the forefront of global efforts to increase access to testing by strengthening laboratory capacity—including through quality management systems—providing training, generating evidence for policy change and exploring innovative delivery models.


Efforts to boost local manufacturing capacity of COVID-19 rapid diagnostic tests (RDTs) helped to drive down prices from US$5.00 to US$2.50, and supported savings of over US$130 million in test procurement in 2021.
We partnered with Unitaid to deliver US$ 50 million in grants that adapt new, LMIC-centred strategies to fill COVID-19 testing and treatment gaps, generating data for global scale-up and simplifying the path to health.

We worked with ACT-Accelerator partners to deliver 147 million cumulative tests to LMICs and trained 17,000 people across 180 countries in the use of antigen RDTs.
WATCH THIS VIDEO TO DISCOVER OUR INTERACTIVE SARS-COV-2 TEST TRACKER
Through its network of qualified partner sites, FIND independently evaluated more than 100 COVID-19 tests in 2021, becoming a trusted source of COVID-19 test data, including documenting the impact of variants on test performance. We made these data available through interactive tools such as our COVID-19 test directory, genomic sequencing capacity map, testing policies map and testing data dashboard.
INVESTING AND PARTNERING FOR GLOBAL IMPACT IN THE PANDEMIC RESPONSE





Championing global manufacturing equity, FIND made multiple investments in technology transfer and local production to boost local COVID-19 RDT manufacturing in LMIC settings worldwide (Brazil, India, Senegal, South Africa).
We laid the groundwork for the uptake of self-testing, including conducting studies that provided the first-ever evidence on feasibility and acceptability of self-testing in LMICs (in Zimbabwe and Malawi). We also assessed values and preferences for self-testing, as well as potential demand in eight LMICs.
Supporting early detection and rapid response, FIND mapped next-generation sequencing of SARS-CoV-2 globally, supporting LMICs to considerably expand genomic surveillance capacities. FIND-funded projects drove the identification of the Omicron variant in Botswana, for example, and support "MicroLabs" in India.


Our ACT-Accelerator partnership with the International Committee of the Red Cross (ICRC) focused on testing among vulnerable populations in complex emergencies and conflict areas (Palestine, Eastern Democratic Republic of the Congo).

A collaboration with the International Olympic Committee on testing strategy for the Japan 2021 Summer Olympics helped strengthen health policies for sporting events.
"COVID-19 has demonstrated that with coordination, focus and funding, what once was considered impossible is now within reach. We are committed to building on the gains that have been accelerated by the pandemic to transform our preparedness and prevent the next one."
DANIEL BAUSCH, SENIOR DIRECTOR, EMERGING THREATS & GLOBAL HEALTH SECURITY
CURBING DRUG RESISTANCE TO PROTECT HARD-WON TREATMENTS
LEARN ABOUT THE NEW RAPID TEST FOR GONORRHOEA DEVELOPED WITH PARTNERS

Together with partners, FIND made progress on the development of a new rapid test for gonorrhoea that helps differentiate it from chlamydia, which will help combat antibiotic overuse and antimicrobial resistance (AMR) in LMICs through accurate diagnosis.

As a result of a successful call for proposals for the development of diagnostic tools that meet the technical characteristics of a target product profile we developed, FIND is partnering on a new project to evaluate and develop new blood culture technology adapted for use in LMICs, the results of which will be available at the end of 2022.

DRIVING DIAGNOSTIC INNOVATION IN SUPPORT OF universal health coverage (uhc)
While the global response to COVID-19 has accelerated diagnostics development and access, it is also clear that the pandemic has slowed—and in some cases reversed—gains made against diseases such as malaria and tuberculosis (TB). Testing is the cornerstone of UHC, requiring us to work together to apply the diagnostic innovation spurred on by the pandemic to address primary healthcare more broadly.
"Diagnostic gaps are stymieing global health targets for too many infectious and non-communicable diseases. We are working across programmes and with partners to ensure the right test is available and affordable for the people who need it, whenever they need it, wherever they are. Today we are faced with unprecedented opportunities to better serve and improve the health of patients and communities around the world."
SERGIO CARMONA, CHIEF MEDICAL OFFICER
HEAR PERSPECTIVES FROM OUR PARTNERS ON
UNIVERSAL HEALTH COVERAGE DAY 2021
FEVER AND MALARIA


FIND invested in two companies to develop novel, point-of-care platforms to perform complete blood counts and triage severe infections at the primary care level.
We enrolled 5,700 participants in the AMR Diagnostic Use Accelerator study, our flagship study in Africa and Asia to address fever management, the appropriate use of antibiotics at primary care level, and to support healthcare workers to detect severe infections for timely referral.
Hepatitis


We worked to expand access to testing for hepatitis C virus (HCV) to key populations and vulnerable groups, driving the generation of robust evidence to demonstrate the cost-effectiveness and health impact of HCV test-and-treat programmes and informing groundbreaking new WHO guidance on HCV testing and uptake in multiple countries.
Implementation of an HCV testing and care model in prisons in India led to access to HCV services for 70,000 inmates.
COMMUNITIES FROM AROUND THE WORLD SHARE PERSPECTIVES ON HCV SELF-TESTING
Neglected Tropical Diseases (NTDs)

FIND supported Kenya in the final steps of disease elimination for human African trypanosomiasis (HAT), which is currently being submitted to WHO for validation.
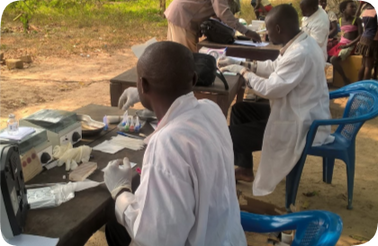

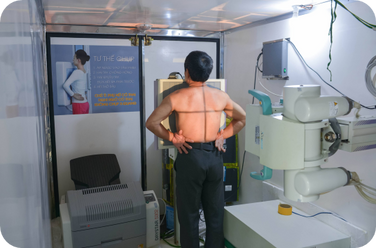

tuberculosis



In 2021, our investments in new TB diagnostics paved the way for new innovations, including CAD-based imaging, point-of-care molecular platforms, and next-generation LAM testing. We also established a catalogue of mutations that predict drug resistance in TB strains.
We secured a new Global Fund grant for TB testing, with forays into testing linked to TB preventive therapy and accelerating TB elimination in India.
Through the Joint Effort for Elimination of Tuberculosis (JEET) project—the largest private-health-sector engagement initiative for TB ever carried out in India—we notified more than 200,000 individuals that they had TB and over 80% of them were successfully treated.

"Testing is the eyes and ears of every country’s health system, providing decision-makers with data on everything from the impact of public health measures to the emergence of new COVID-19 variants. Strengthening diagnostic capacity so that national targets can be met for all priority diseases depends on trusted relationships and actively listening to countries' needs for everything we do."
SANJAY SARIN, VICE PRESIDENT, ACCESS
Innovating fit-for-purpose technology to optimize impact across diseases






FIND invested US$21 million to advance decentralized point-of-care molecular testing. Four new platforms in late-stage development represent the next wave of disruptive molecular testing platforms for use in pandemic preparedness, AMR, TB, fever management and disease surveillance.
We helped expand access to testing through five COVID-19 digital data capture tools, developed and piloted by FIND, which increased access to COVID-19 testing for commuters, drivers and vendors in taxi ranks, as well as among community health workers.
Digital tools also enabled bi-directional testing services for TB and COVID-19 among motorcycle transport providers.
Following completion of a successful pilot, we have received a new grant to expand local capacity building for diagnostic network optimization (DNO) across 15 LMICs over the next three years and launched the OptiDx microsite to support in-country deployment.

"Global momentum is driving innovation that goes beyond a single disease, from digital technologies to self-tests and local manufacturing expansion. Investing in the right technology can transform access to testing across all diseases—so that performing a test, interpreting the result and connecting someone to the right care is simple and accurate every time, in every setting."
MARTA FERNáNDEZ SUáREZ , CHIEF TECHNOLOGY OFFICER

WORKING WITH PARTNERS TO
ADVANCE TESTING FOR ALL
We work in more than 70 countries, through both local and regional partnerships. Hear from some of our partners and from communities around the world.
case-finding of
non-communicable diseases in India
Schistosomiasis detection in Kenya
COVID-19 antigen rapid diagnostic tests in Ethiopia

Testing must be an integral part of sustainable, resilient health systems worldwide to accelerate progress towards UHC and global health security. The past year is proof that the world has never been more ready to work together to turn promising innovation into concrete improvements and better community health outcomes across all diseases. We must stop the cycle of “panic then forget” once and for all.

KEEPING TESTING HIGH
ON THE GLOBAL AGENDA
"The pandemic has shown just how fundamental testing is to well-functioning health systems. Our challenge now is ensuring that it stays on the agenda so that countries can put testing at the heart of their national health strategies as they build back better. The continued support of our donors and partners is critical at this pivotal moment."
WILLO BROCK, VICE PRESIDENT, EXTERNAL AFFAIRS
launch of THE FIND 2021 strategy and
high-level forum on diagnostic testing

LAUNCH OF THE FIND 2021 STRATEGY AND HIGH-LEVEL FORUM ON DIAGNOSTIC TESTING
Hear from distinguished guests how COVID-19 has transformed testing, the implications for diagnostics in health emergencies and UHC, and how this has informed the FIND 2021 strategy.

ENGAGEMENT IN
G7/G20 MEETINGS
FIND played a critical role in the development of initiatives for pandemic preparedness, including guiding plans for the testing strategy of the G7 100 Days Mission in partnership with CEPI.

CALL TO ACTION FOR WORLD LEADERS
More than 40 organizations, leaders and advocates signed a call to action issued by FIND ahead of the G20 health meetings, calling for world leaders to uphold diagnostics and equitable access to testing within their commitments to support sustainable recovery from COVID-19.

THE LANCET COMMISSION ON DIAGNOSTICS
The Lancet Commission on Diagnostics set out a roadmap of 10 key recommendations to help stem inequities in access to tests and quantified the impact that could be achieved by reducing diagnostic gaps. The study was co-authored by the CEO of FIND, and we co-hosted the global launch event.
global launch of the lancet commission on diagnostics
THANK YOU TO OUR DONORS
AND ALL OUR PARTNERS
















Our work is made possible by our donors, alongside significant financial contributions from our private sector partners.

Photo credits:
Cover: FIND & MYRADA/Adithya Shanka
Table of contents: FIND/George Muiruri
Introducing and scaling...: Abdul Razak Abdul Latif/Dreamstime.com
Investing and partnering...: Jakayla Toney/Unsplash; Robert Kneschke/Dreamstime.com; FIND; Abed Rahim Khatib/Dreamstime.com; Canva
Neglected tropical diseases: FIND/J.Ndung'u
Tuberculosis: Friends for International Tuberculosis Relief (FIT) Viet Nam
Innovating fit-for-purpose...: Friends for International Tuberculosis Relief (FIT) Viet Nam
Keeping testing high on the ...: FIND/George Muiruri
Closing picture: FIND & MYRADA/Shivram Manthena